This post includes the full notebook for Sept-Dec 2022 Acropora pulchra adult coral experiment Moorea, French Polynesia. These notebook posts are updated in order of most recent to oldest.
Project Overview
The goal of this project is to expose adult Acropora pulchra corals to conditions that elicit changes in symbiotic and metabolic condition and test how these changes influence response to a repeat stressor. Here, we are manipulating light and feeding conditions to elicit metabolic and symbiotic responses and testing stress response at the end of the experiment using the thermal stress CBASS system. The team will expose adult corals to light with added food, light with no added food, and shade with added food in Sept - Dec with thermal stress phenotype measurement in Dec with S. Matsuda and R. Cunning.
The experimental design overview can be seen here:
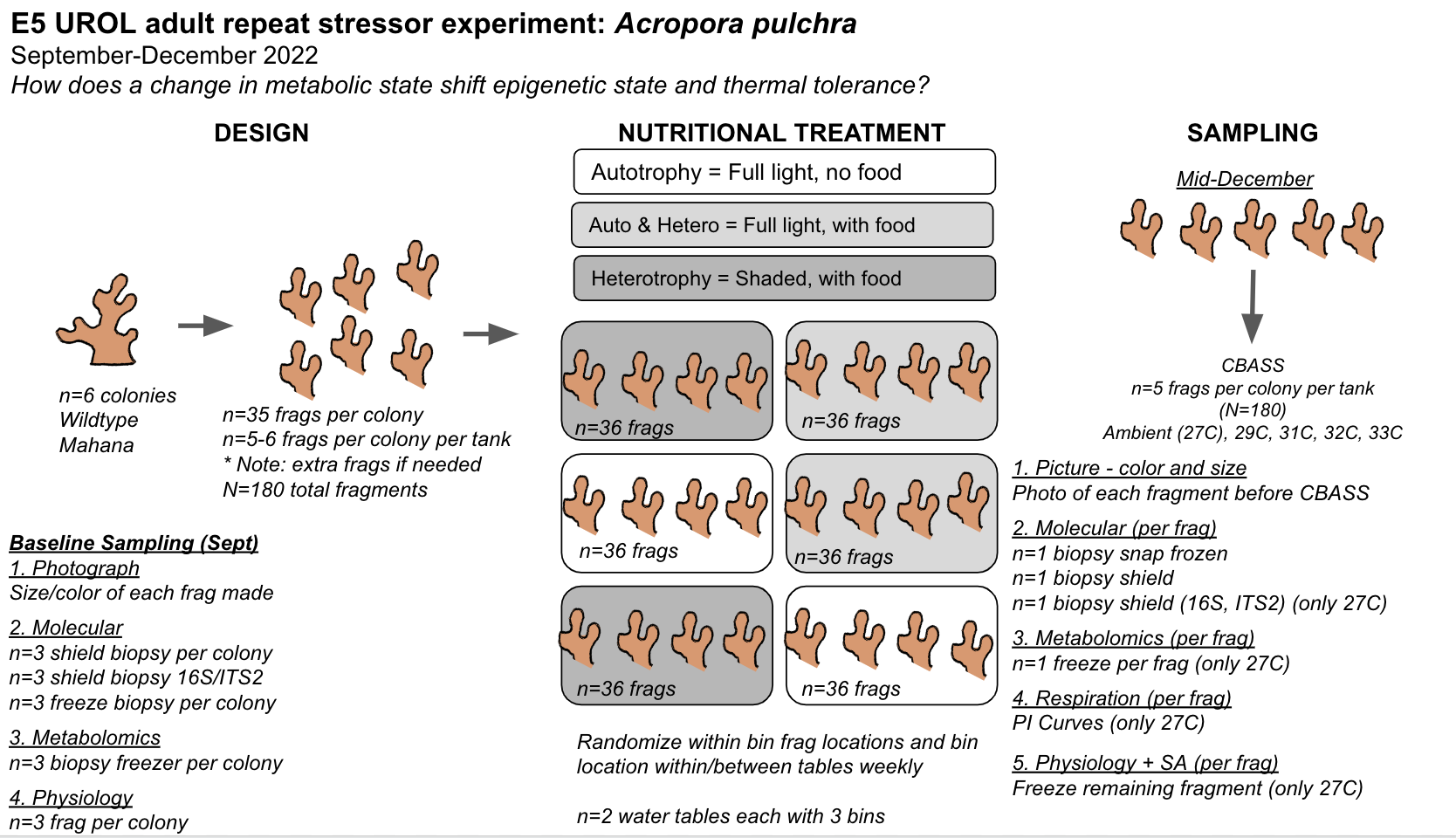
This post details activities and notebooks for this project.
Field expedition members:
Ariana Huffmyer, URI
Pierrick Harnay, URI
Danielle Becker-Polinski, URI
Hollie Putnam, URI
Shayle Matsuda, Shedd Aquarium
Ross Cunning, Shedd Aquarium
Helpful links
The following protocols and links to data will be used regularly by the field team conducting this project.
Daily measurements:
Daily measurements protocol
Daily measurement data sheet
Schedule daily checklist
Weekly maintenance:
Weekly maintenance protocol
Schedule weekly checklist
Fragment metadata and sample tracking:
Fragment tracking datasheet
Sample inventory
Baseline sampling:
Baseline sampling protocol
Baseline sampling checklist
Sample inventory
Logger files:
Logger files folder
Feeding protocol:
Feeding protocol
Final sampling:
Final sampling protocol
Final sampling checklist
Sample inventory
Fragment tracking datasheet
26 November 2022
Daily flow checks
Checked flows throughout the day 3 times.
25 November 2022
Daily measurements
Cleaned all tanks in the afternoon. Cleaned all tubing lines and flushed the main line to improve flow. Downloaded temp/light loggers. One overgrown coral F16 was removed from tank 4. Fed corals from 18:00- 18:30
Light
24 November 2022
Daily flow checks
23 November 2022
Daily flow checks
22 November 2022
Planning final sampling
Hollie, Ariana, Shayle, and Ross started planning the final sampling for this experiment. Ariana drafted sampling checklist and protocols for the team to revise and discuss over the next couple of weeks before sampling.
Final sampling protocol
Final sampling checklist
Final sample inventory
Fragment tracking datasheet
21 November 2022
Daily measurements
20 November 2022
Daily measurements
19 November 2022
Daily measurements
18 November 2022
Daily measurements
17 November 2022
Daily measurements
16 November 2022
Daily measurements
15 November 2022
Daily measurements
14 November 2022
Daily measurements
Hollie and Pierrick took daily measurements between 18:30 - 18:45.
13 November 2022
Putnam cleaned all corals and tanks.
Daily measurements
12 November 2022
Daily measurements
11 November 2022
Daily measurements
Daily measurements were taken starting at 16:32.
10 November 2022
Daily measurements
Daily measurements were taken starting at 16:28.
09 November 2022
Daily measurements
08 November 2022
Daily measurements were taken starting at 13:30.
Daily measurements
07 November 2022
Daily measurements
Daily measurements were taken starting at 13:20.
06 November 2022
Daily measurements
Hollie and Pierrick took daily measurements between 17:45 - 18:00. The heaviest of rains have ended, but cloud cover is still high and salinity is decreasing.
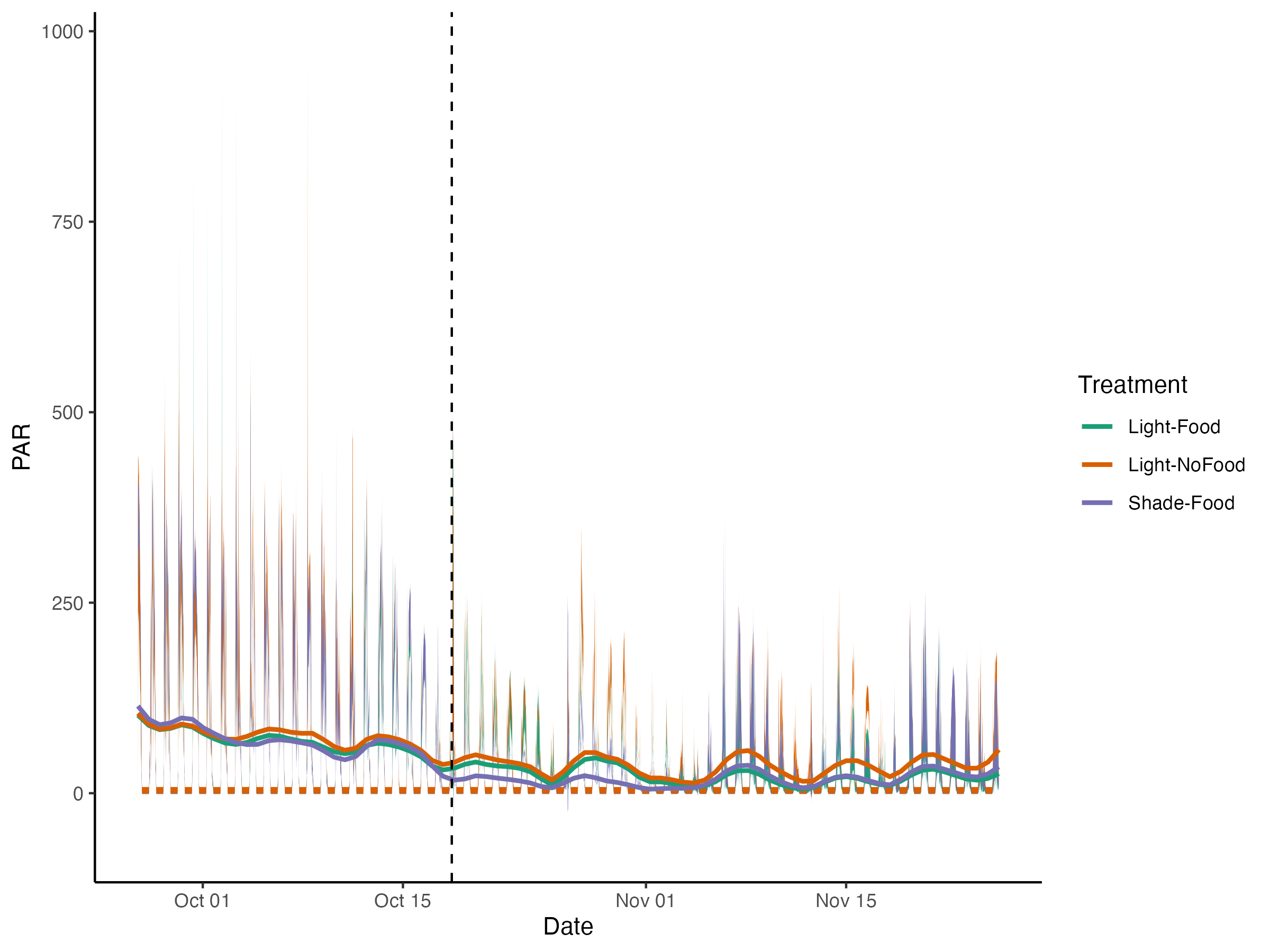
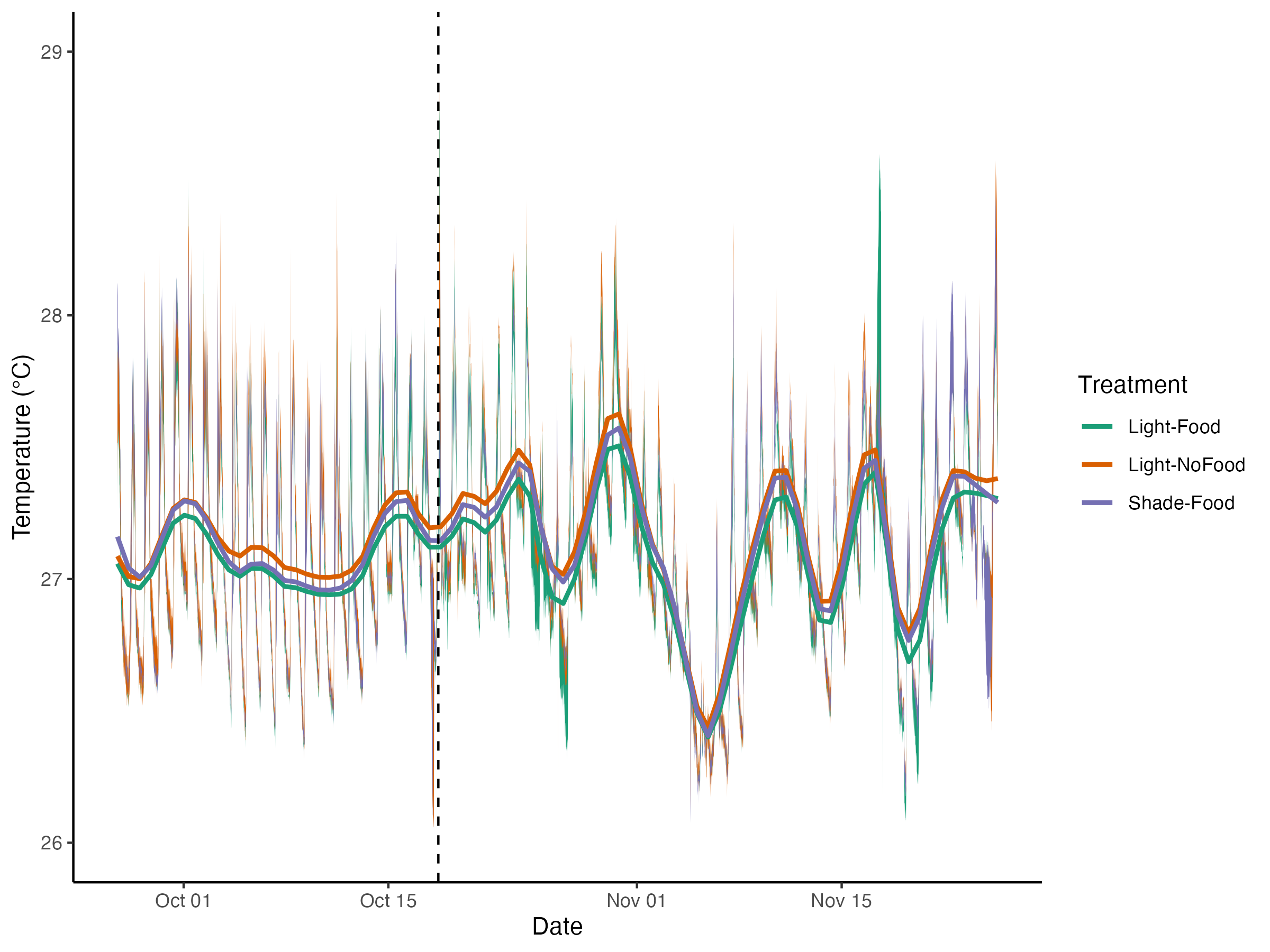
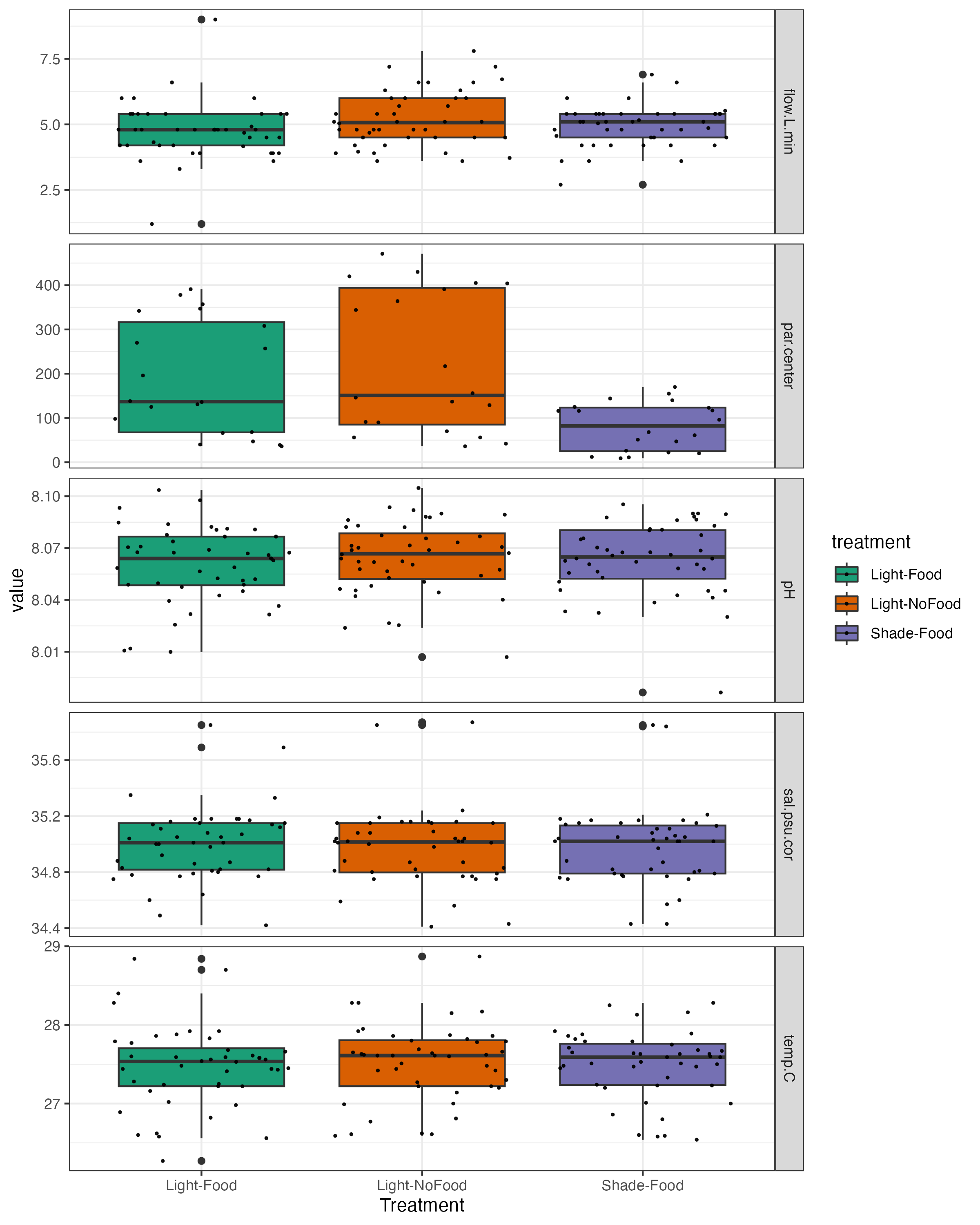
Hollie entered data, updated daily measurment plots and pushed to github.
The plots of daily measurements by date and tank.
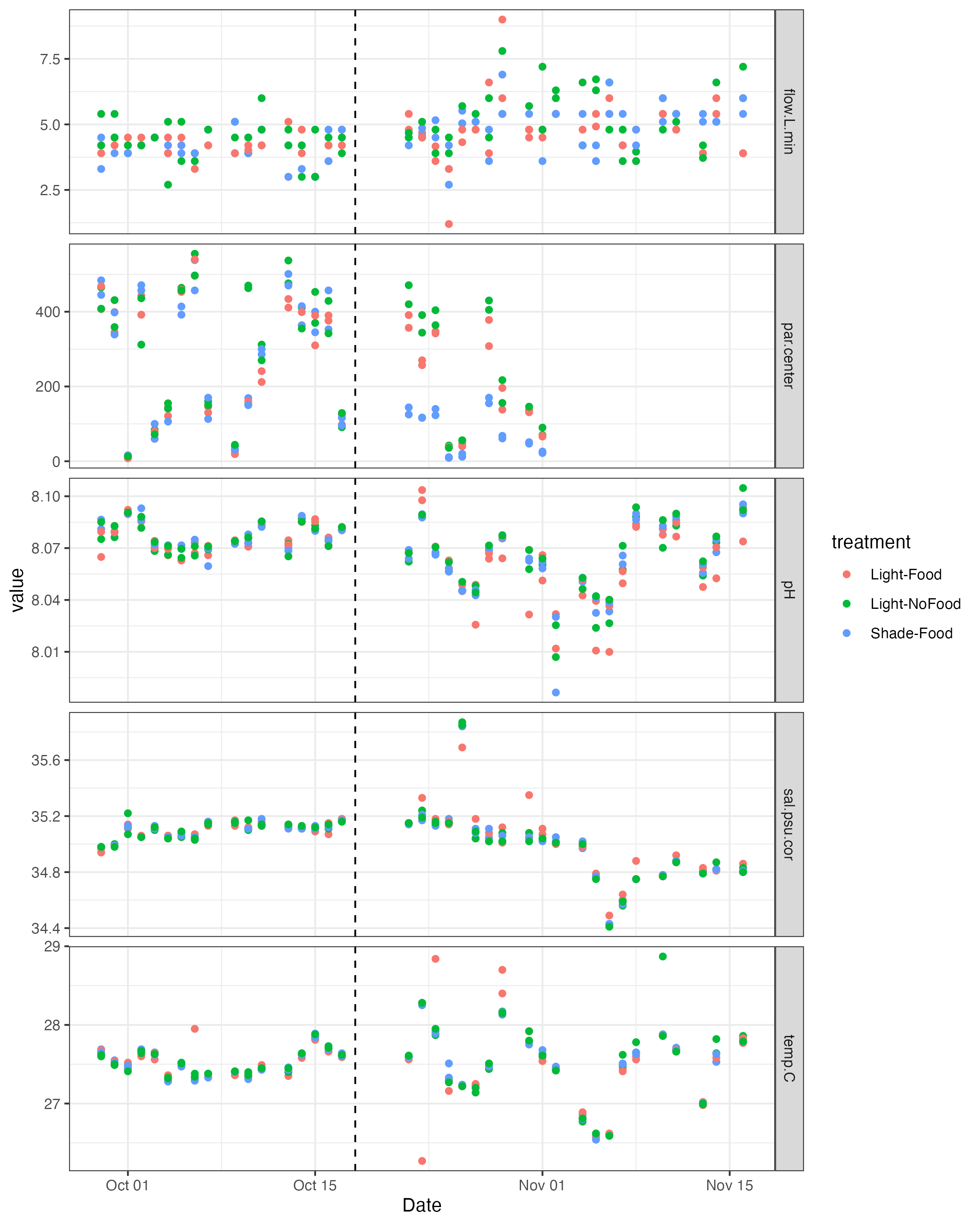
The plots of environmental variables by treatment.

####ANOVA of light from the exposure period ####Light
summary(aov(par.center~treatment, data=daily_calc_exposure))
||||||| |—|—|—|—|—|—| | |Df| Sum Sq| Mean Sq| F value | Pr(>F) | treatment | 2| 217323 | 108662 | 7.249 |0.00157 | Residuals | 57 |854426| 14990 | | |
05 November 2022
Daily measurements
Hollie and Pierrick took daily measurements between 16:00 - 16:15. Heavy rains continue.
Apogee light meter got moisture inside and is drying. Hollie replaced the battery.
04 November 2022
Daily measurements
Tanks and coral strings were all cleaned between 14:00 and 16:00. Putnam and Harnay completed daily measurements between 16:16 and 16:30. Corals were fed 16:35 - 17:05.
Tank1

Tank2

Tank3

Tank4

Tank5

Tank6

03 November 2022
No Activity.
02 November 2022
Daily measurements
C31 was identified as dead and removed from tank 6. Putnam and Harnay completed daily measurements between 17:45 and 18:00.
01 November 2022
Daily measurements
Terava completed daily measurements between 13:15 - 13:40.
31 October 2022
Daily measurements
Terava completed daily measurements between 13:00 - 13:30.
30 October 2022
No activity.
29 October 2022
Daily measurements
Daily measurements were collected by Terava at 14:30 - 14:40.
28 October 2022
Daily measurements
Daily measurements were collected at 12:20 - 12:30.
27 October 2022
Daily measurements
Daily measurements were taken at 18:00 - 18:10.
26 October 2022
Daily measurements
Daily measurements were taken at 09:20 - 09:30.
Cleaning tanks
Terava and Danielle cleaned the E5 tanks today from 12:00 - 16:00.
25 October 2022
Daily measurements
Daily measurements were taken at 15:00 - 15:10.
24 October 2022
Daily measurements
Daily measurements were taken at 09:15 - 09:20.
23 October 2022
Daily measurements
Danielle and Hollie took daily measurements today of the E5 tanks and the squaricals between 1500 and 1700.
22 October 2022
Daily measurements
Hollie and Danielle completed daily measurements and cleaned the strings for all of the fragments. During spawning we are also taking daily measurements of squaricals and buckets for reference.
Daily measurements are recorded in GitHub here
21 October 2022
Daily measurements and feeding
Daily measurements were taken in E5 tanks, parent tanks, and larval tanks today between 1530-1630. Daily measurements are recorded in GitHub here. We fed the E5 corals today following the E5 A. pulchra metabolism feeding protocol here. Feeding occurred from 1545-1645.
20 October 2022
Daily measurements
Daily measurements were taken in E5 tanks, parent tanks, and larval tanks today between 1600 and 1700. Daily measurements are recorded in GitHub here.
19 October 2022
Embryo rearing
Embryos from 10/18 spawning were checked and stirred every 2-3 hours today. We also frequently check the fill level of the coolers. Due to high water flow and limitations in rate of filtering in the filter line, the coolers will sometimes drain which risks damaging the pump and stopping water flow. We closely monitor the level of the coolers every 2-3 hours. 50 um in line filters were rinsed and cleaned every 4-7 hours. The coolers were also cleaned today.
Overall, embryos from 10/17 and 10/18 spawning are doing well with minimal mortality.

Feeding
We fed the E5 corals today following the E5 A. pulchra metabolism feeding protocol here. Feeding occurred from 16:35-17:35.
Daily measurements
Daily measurements were taken in E5 tanks, parent tanks, and larval tanks today between 1430 and 1500. Daily measurements are recorded in GitHub here.
In Tank 6, C31 is showing high partial mortality (~90%) with algal overgrowth.
18 October 2022
Cleaning tanks
We cleaned all tanks for the E5 experiment today. All fragments look healthy and no mortality was observed.
Loggers
All E5 loggers were downloaded between 1200 and 1400. The logger data was ran using the logger R script in the GitHub repo. Our environmental data looks like this:

17 October 2022
Daily measurements
Ariana and Terava took daily measurements today between 12:45 and 13:15. Daily measurements are recorded in GitHub here. Tanks will be cleaned tomorrow.
E5 experiment - symbiont counts
Ariana and Terava finished the symbiont density measurements today. The proceedure followed the protocol for E5 processing with no modifications. Data is recorded in GitHub.
Ariana also ran the data using R scripts to analyze data as we go based on the E5 timeseries project scripts.
The symbiont cell density data by colony/genotype looks like this:
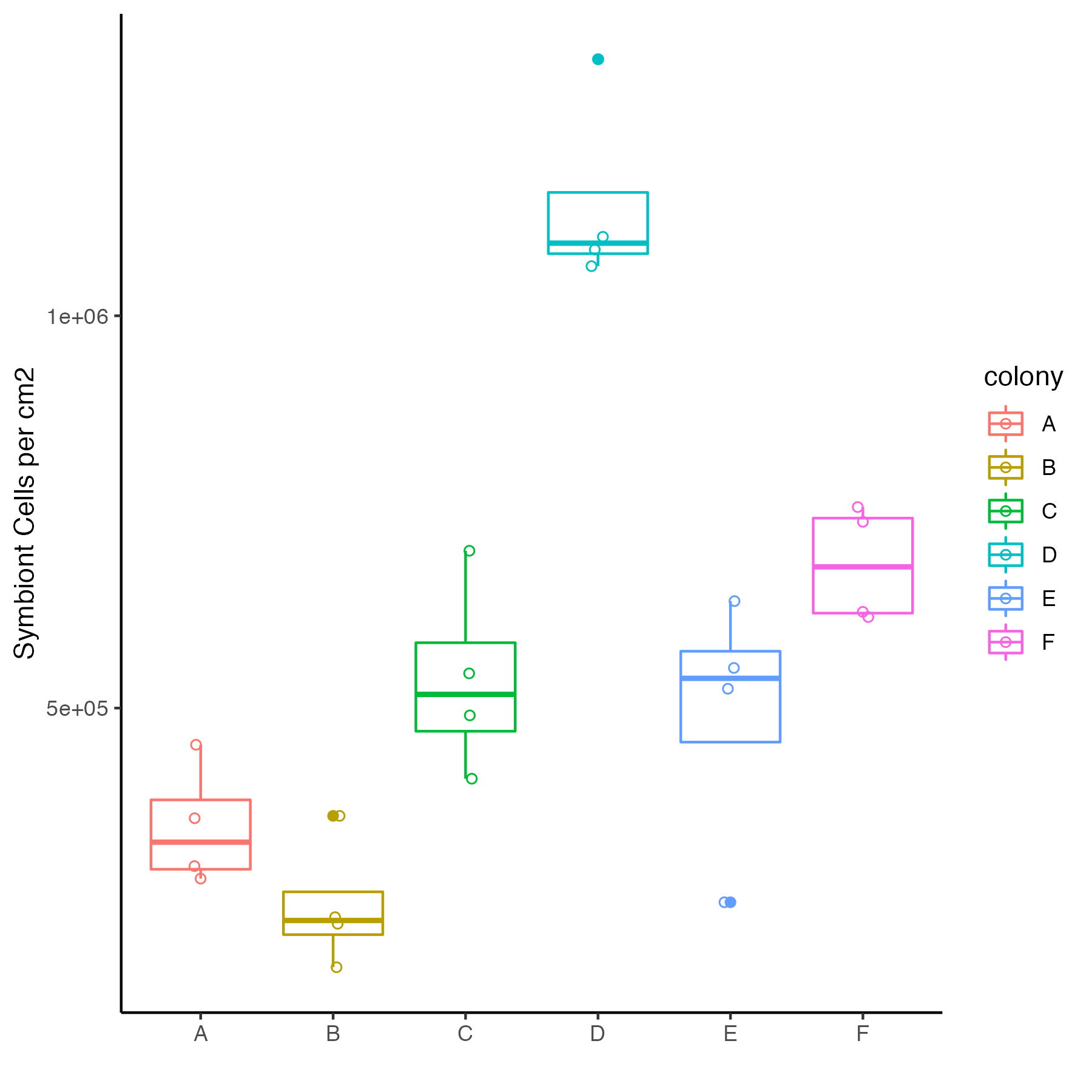
There is a significant difference in symbiont density between colonies.
lmer(cells.cm2~colony + (1|tank), data=.)
p<0.001
E5 experiment - starting treatment
Today we are starting treatments in our E5 A. pulchra metabolism experiment. The treatments for each tank are as follows:
| Tank | Light Treatment | Food Treament |
|---|---|---|
| 1 | Full light | Added food |
| 2 | Full light | No food |
| 3 | Shaded | Added food |
| 4 | Full light | Added food |
| 5 | Shaded | Added food |
| 6 | Full light | No food |
Calculating the amount of food needed
For Coral Max food, the manufacturers instructions suggest 1/2 teaspoon per 50 gallons of tank water. Our tanks are 19.3 gallons (29 cm x 60 cm x 42 cm). Therefore, we would need 0.368 of a 1/2 teaspoon dose (19.3 / 50 gallons).
The mass of 1/2 teaspoon (converted to volume of 2.464 mL) dose is 1.148 g. 1.148 g * 0.368 = 0.422 g. 0.422 g is the amount we need for the size of our tanks. We will round up. For each tank, we will add 0.5 g of food at each feeding.
If we feed 3 times per week in each of 4 tanks (those that have food in the treatment), then we will use 6 g per week. If we run the experiment for 8 weeks, we will need a total of 48 g. We have >200 g, so we have plenty for this experiment.
We then weighed 0.5g into a 5 mL tube and marked the level that the food was at. 0.5g of food is exactly at the 1 mL mark on the white cap 5mL tubes.
We labeled 4 5mL tubes with the tank number for all tanks that recieve food (1, 3, 4, and 5) and marked with a sharpie the 1mL mark to fill food. At each feeding, the person feeding will fill tube to this mark and then add to the tank. See the feeding section below for a protocol on feeding.

E5 experiment - adding shade
We added shade cloth to tanks 3 and 5 at 13:09 with one layer of shade cloth. We recorded light in the tanks after shade cloth was added. Daily measurements were taken before shade cloth was added. The values of light are included below that show the comparison of light before and after shading in each tank.
Light before adding shade:
| Tank | Time | Center | Q1 | Q2 | Q3 | Q4 |
|---|---|---|---|---|---|---|
| 3 | 1249 | 96 | 120 | 118 | 116 | 115 |
| 5 | 1252 | 116 | 103 | 102 | 83 | 90 |
Light after adding shade:
| Tank | Time | Center | Q1 | Q2 | Q3 | Q4 |
|---|---|---|---|---|---|---|
| 3 | 1309 | 59 | 65 | 56 | 65 | 53 |
| 5 | 1310 | 38 | 36 | 31 | 38 | 32 |
Light is reduced by approx. 35-55% after adding shade.

E5 experiment - feeding
The process for feeding is as follows and is described in the E5 A. pulchra metabolism feeding protocol here. The overview of the steps are as follows:
- Measure out food as described above into labeled 5 mL tubes to the marked 1 mL mark with Coral Max.
- Turn off water on the main line to all tanks.
- Use the nalgene bottle or a tri pour to take ~ 1 L water out of the tanks to ensure the food doesn’t overflow.
- Add the food to each respective tank (tanks 1, 3, 4, 5) and stir and break up the chunks with a transfer pipette.
- Start a timer for 1 h.
- After 1 h, turn the water flow back on.
Repeat this process 3 times per week (e.g., Mon, Wed, Fri).
Record the timing and notes about feeding in the schedule tracking document here.
Today coral feeding started at 13:53 and water was turned on at 14:55.
16 October 2022
Hollie arrived today and joined our field team!
Daily measurements
Ariana and Danielle took daily measurements in the E5 experimental tanks as well as the tanks holding adult colonies between 1345 and 1415. Daily measurements are recorded in GitHub here.
15 October 2022
Daily measurements
Ariana took daily measurements in the E5 experimental tanks as well as the tanks holding adult colonies between 1400 and 1430. Daily measurements are recorded in GitHub here.
E5 experiment - symbiont counts
Ariana counted symbiont density in more samples today. The proceedure followed the protocol for E5 processing with no modifications. Data is recorded in GitHub.
Ariana also wrote scripts to analyze data as we go based on the E5 timeseries project scripts.
Preliminary data shows that symbiont densities (~1e+06) are in the range seen in the E5 timeseries project.

The remaining samples will be processed over the next few days.
Tank cleaning
We flushed all water lines to improve water flow at 2100.
14 October 2022
Daily measurements
Ariana and Teravatook daily measurements in the E5 experimental tanks as well as the tanks holding adult colonies between 1000 and 1100. Daily measurements are recorded in GitHub here.
Symbiont counts
Ariana and Terava started symbiont cell density measurements on the symbiont fraction of samples that were separated yesterday. The proceedure followed the protocol for E5 processing with no modifications.
Data is recorded in GitHub. Five samples were analyzed today. The remaining samples will be processed over the next few days.
Analyzing data
Ariana wrote scripts to analyze protein and surface area for the baseline E5 adult measurements. The scripts are located on GitHub.
Protein measurements from yesterday were briefly analyzed. These are the protein values for each colony/genotype in the host and holobiont fraction in the figure below.
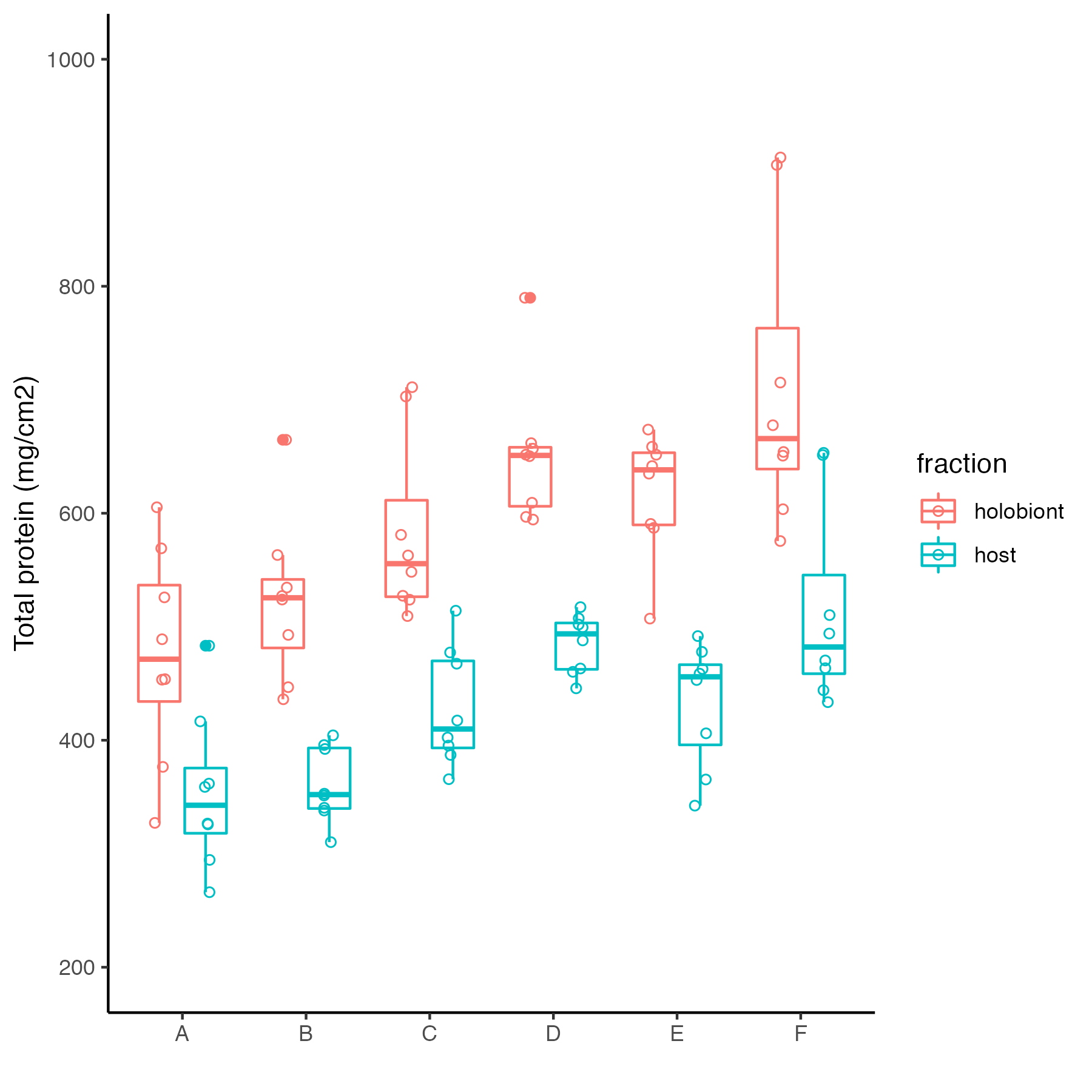
A linear mixed model testing differences in protein by genotype show a significant difference in both host and holobiont protein by genotype.
lmer(protein_ug.cm2~colony + (1|tank), data=protein)
13 October 2022
Daily measurements
Ariana took daily measurements in the E5 experimental tanks as well as the tanks holding adult colonies between 1030 and 1100. Daily measurements are recorded in GitHub here.
Baseline samples - total protein
Ariana, Danielle, and Terava conducted total soluble protein assays (Pierce BCA protein assay) for host and holobiont fractions for Danielle’s histology time series samples and the E5 adult baseline samples following the E5 total protein protocol.
The summary of the protocol process for Ariana’s samples was as follows. Danielle and Ariana followed the same protocol with an added step for host-symbiont fraction separation for Ariana’s samples. 2 plates were run for host samples and 2 plates were run for holobiont samples in total.
- Thaw two 1mL tube samples for each E5 fragment.
- For one of the tubes, we separated host and symbiont fraction. The second tube will have the holobiont fraction.
- One tube per fragment was thawed and centrifuged at 3500 rpm for 3 min using the Fisher centrifuge as used during symbiont isolation trials on 3 Oct 2022.
- The supernatant (host fraction) was transfered to a new tube labeled with the fragment ID and “H” for “host”. The tube with the symbiont pellet was labeled with an “S” for “symbiont”.
- For each sample, this resulted in one tube with holobiont tissue, one tube with host tissue, and one tube with symbiont pellet.
- The symbiont fraction was returned to the freezer at -40C for symbiont counting tomorrow.
- Standards and working solution were generated as described in the total protein protocol.
- First, host fraction samples were run by loading 25 uL of each sample in duplicates in a 96 well plate along with duplicates of each standard. The plate map for host samples is below.
- The plate was gently rocked by hand to mix for 30 sec and then incubated at 37°C for 30 minutes.
- After incubation, the plate was allowed to cool at room temperature for 6 min.
- The absorbance of the plate was read on the microplate spectophotometer reader at 562 nm and saved.
- In the next plate, holobiont samples were added in duplicates with 25 uL of each sample and each standard. The plate map is included below.
- The plate was incubated and read in the same way as described above.
- All plates and supplies were cleaned following plate reading.
Host samples were added to the incubator at 1313 and read at 1351. Holobiont samples were added to the incubator at 1356 and read at 1430.
A host and holobiont plate was run prior to these samples by Danielle for the gametogenesis time series.
The data was saved and added to GitHub here.
Plate Map Host (E5 samples):
| Row | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | STND-A | STND-A | STND-B | STND-B | STND-C | STND-C | STND-D | STND-D | STND-E | STND-E | STND-F | STND-F |
| B | STND-G | STND-G | STND-H | STND-H | STND-I | STND-I | A7 | A7 | A16 | A16 | A20 | A20 |
| C | A31 | A31 | B9 | B9 | B20 | B20 | B25 | B25 | B34 | B34 | C1 | C1 |
| D | C19 | C19 | C26 | C26 | C30 | C30 | D4 | D4 | D13 | D13 | D26 | D26 |
| E | D30 | D30 | E5 | E5 | E8 | E8 | E20 | E20 | E31 | E31 | F2 | F2 |
| F | F9 | F9 | F17 | F17 | F22 | F22 | ACR465 | ACR465 | - | - | - | - |
| G | - | - | - | - | - | - | - | - | - | - | - | - |
| H | - | - | - | - | - | - | - | - | - | - | - | - |
Plate Map Holobiont (E5 samples):
| Row | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | STND-A | STND-A | STND-B | STND-B | STND-C | STND-C | STND-D | STND-D | STND-E | STND-E | STND-F | STND-F |
| B | STND-G | STND-G | STND-H | STND-H | STND-I | STND-I | A7 | A7 | A16 | A16 | A20 | A20 |
| C | A31 | A31 | B9 | B9 | B20 | B20 | B25 | B25 | B34 | B34 | C1 | C1 |
| D | C19 | C19 | C26 | C26 | C30 | C30 | D4 | D4 | D13 | D13 | D26 | D26 |
| E | D30 | D30 | E5 | E5 | E8 | E8 | E20 | E20 | E31 | E31 | F2 | F2 |
| F | F9 | F9 | F17 | F17 | F22 | F22 | ACR465 | ACR465 | - | - | - | - |
| G | - | - | - | - | - | - | - | - | - | - | - | - |
| H | - | - | - | - | - | - | - | - | - | - | - | - |
11 October 2022
Daily measurements
Ariana and Danielle took daily measurements of E5 tanks between 1500 and 1530. No mortality was observed. Daily measurements are recorded in GitHub here. Ariana analyzed the daily measurement data in this R script. Total pH was calculating using the Tris calibration from 09/24/22 following the Silbiger Lab protocol and scripts. The data looks like this:
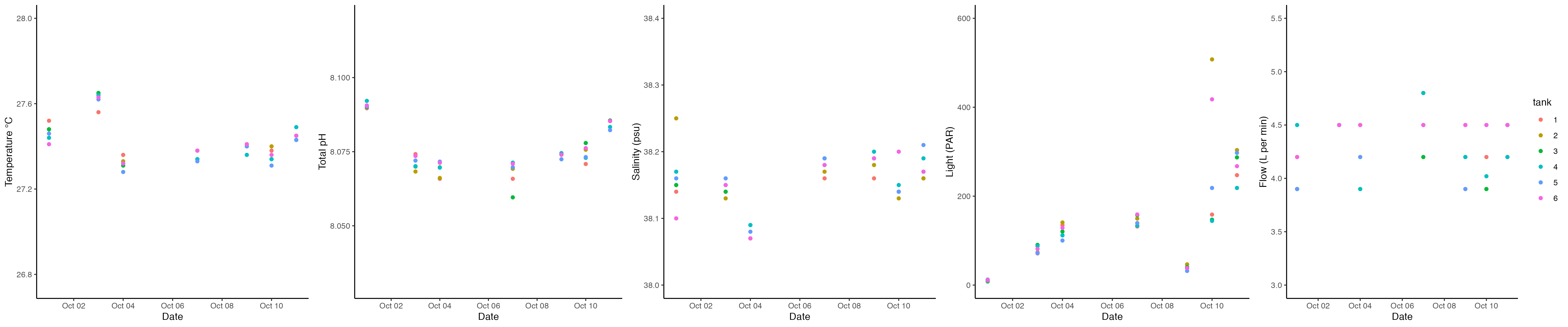
We used one-way ANOVA tests to test for differences in each of these metrics between tanks. There were no significant differences detected.
New coral tags
Ariana continued replacing coral tags for the E5 tanks. All tags were replaced.
Tank cleaning
Ariana and Danielle cleaned all E5 tanks and all corals. Tanks 1, 2, and 3 were moved to a water table further under the shade structure. In the mornings, tanks 1, 2, and 3 have been getting intense morning light at sunrise. Moving the tanks further under the building will make the light more even.

Loggers
Ariana downloaded all Hobo pendant loggers and conducted a preliminary analysis of logger data. We saw that tanks 1, 2, and 3 were getting higher light and temperature peaks in the morning just after sunrise due to the angle of the sun on these tanks. Because of this, we rearranged the tanks to move them under the building structure as described above. The logger data for the E5 tanks is shown below:
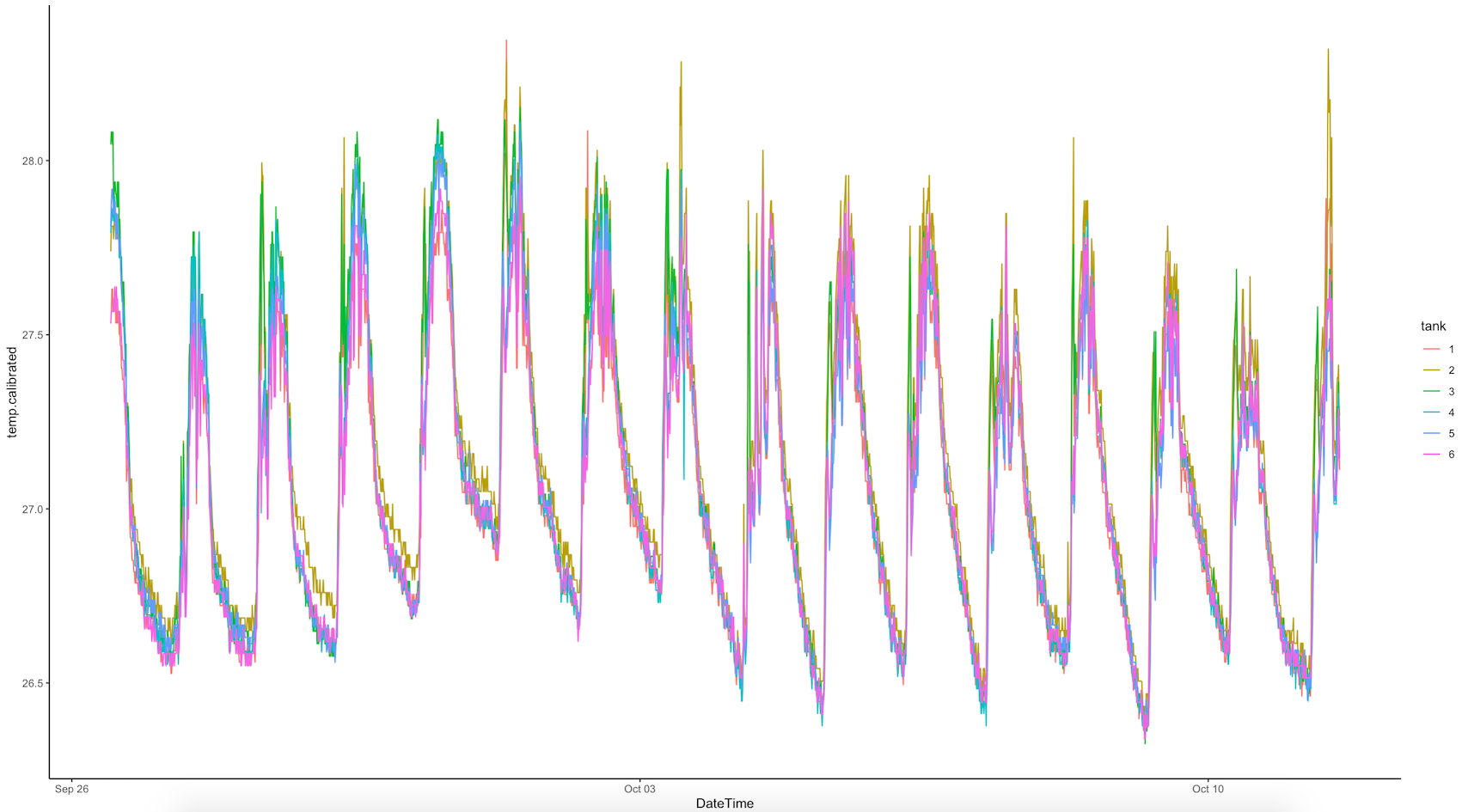
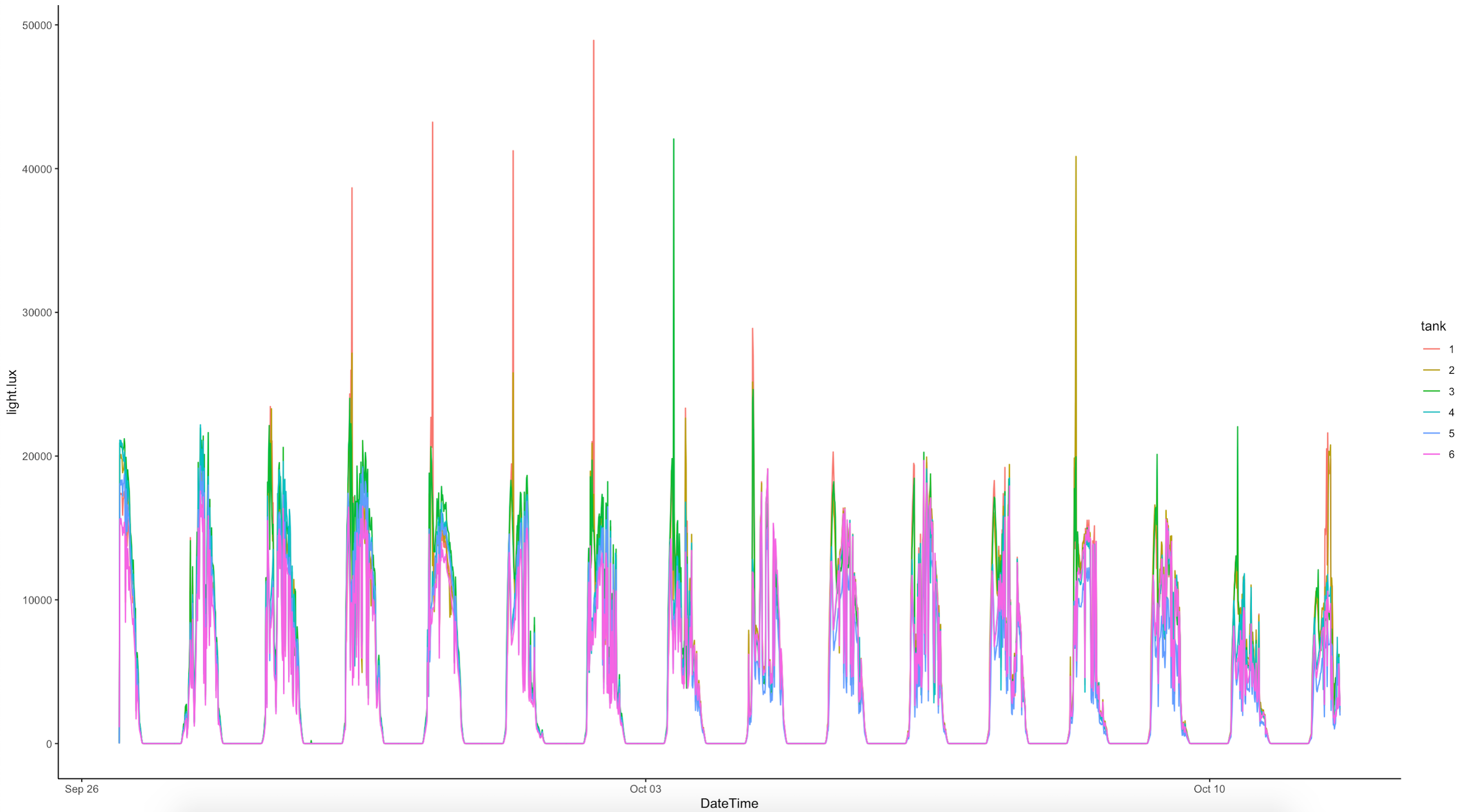
The light measured by the Odyssey loggers is in Integrated PAR, which will be higher values thatn instantaneous PAR measured by the Apogee for daily measurements.
We also plotted the temperatures from the wildtype A. pulchra parent colonies prior to fragmentation.
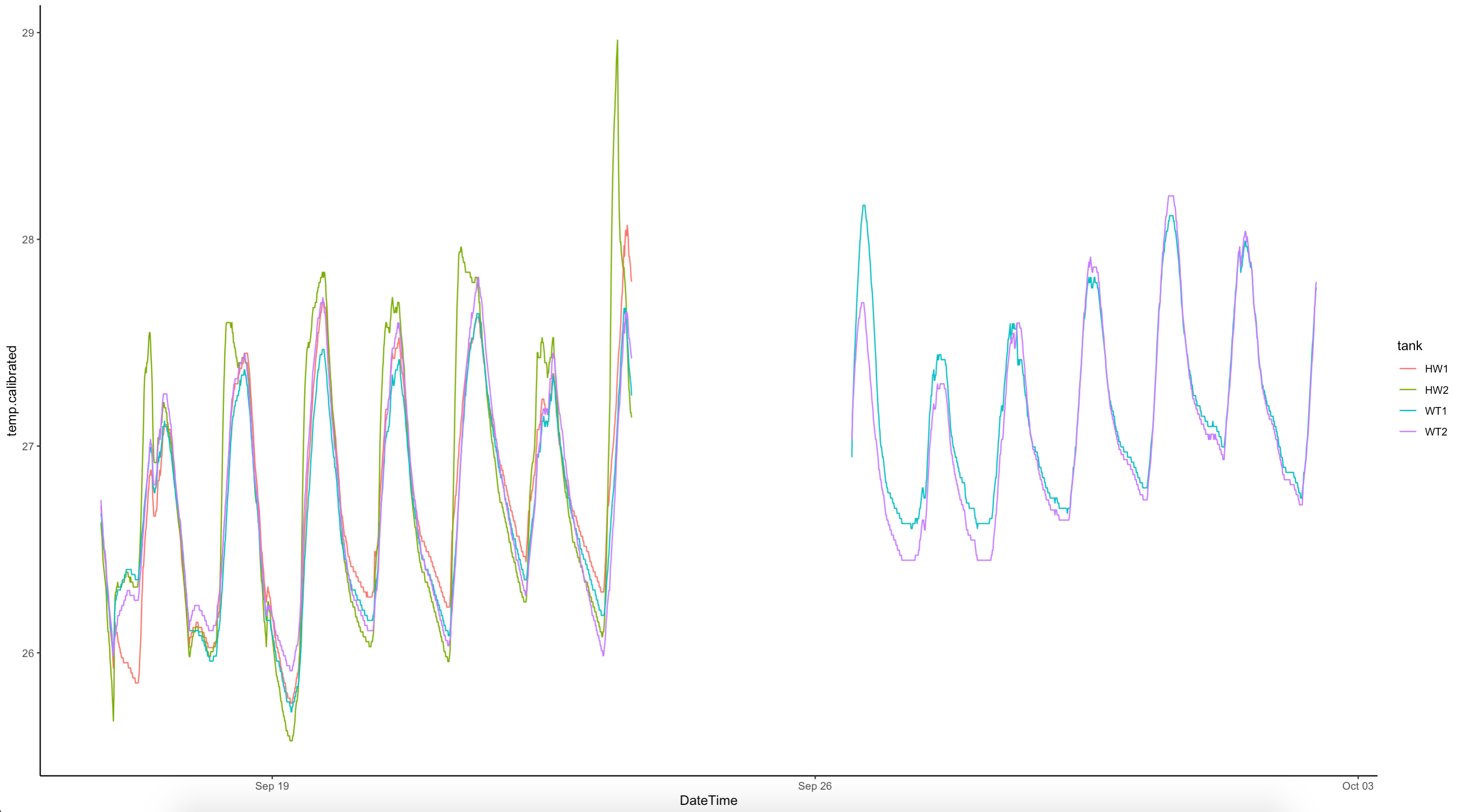
10 October 2022
Today Terava Atger began working with us supported by Danielle’s NSF GRFP funding. Terava will work with us on all projects over the next 3 weeks including the E5 A. pulchra metabolism experiment, histology time series, and coral spawning.
Daily measurements
Ariana and Teravatook daily measurements of E5 tanks between 1000 and 1045. No mortality was observed. Light values were variable during measurements due to passing clouds. Daily measurements are recorded in GitHub here.
New coral tags
Ariana continued replacing coral tags for the E5 tanks.
Aliquoting tissue
Ariana and Terava aliquoted coral tissue slurries for future analysis of symbiont cell counts and total protein. This was completed between 1300-1430. This is the process we followed:
- Thaw a subset of 50mL falcon tubes with frozen tissue slurry, keeping slurry on frozen beads to thaw slowly.
- All homogenate transferred to a labeled 15mL tube. This is used due to low slurry volume in order to homogenize slurry. All 50mL falcon tubes will be cleaned and re used.
- Tissue homogenized for 10-12 sec with the ProScientific PRO200 microtip homogenizer.
- 4 aliquots of 1 mL slurry were added into n=4 snap cap tubes labeled with the fragment ID. In cases where there was not enough to fill all 4 tubes, volumes were noted on data sheet.
- Aliquots were frozen at -40C in a box labeled with “E5 A. pulchra metabolism; Putnam; aliquot tissue slurry; N=24 fragments; n=4 aliquots per fragment”. There are two boxes (Box #3, Box #4).
- This process was repeated for N=24 fragment slurries, with n=96 total tubes with 1 mL aliquots. All remaining slurry in 15 mL tubes was kept in black mesh bag at -40C.
- All equipment was cleaned and sterilized.
The sample tracking sheet can be found here with information on aliquoting sample locations.
9 October 2022
Daily measurements
Ariana took daily measurements of E5 tanks between 1600 and 1630. No mortality was observed. Light values were low in the afternoon due to cloud cover. Daily measurements are recorded in GitHub here.
New coral tags
Ariana started replacing paper clips with coated wire tags. These will be replaced over the course of the next week.
7 October 2022
Daily measurements
Ariana took daily measurements of E5 tanks between 1400 and 1430. No mortality was observed. Daily measurements are recorded in GitHub here.
Surface area measurements
Ariana measured surface area using the wax dipping method on all E5 baseline fragments. All airbrushed fragments from 5 October 2022 were dried in a drying oven at 80°C for 6 hours. The fragments were then weighed on a Mettler Toledo Excellence Plus SP203S scale to 0.001 g. This was recorded as the fragment dry weight. Fragments were then dipped once in melted wax and allowed to dry. Fragments were then re-weighed on the scale and recorded as wax dipped weight.
We will use the standard curve for this wax batch from the one generated by Danielle during this field trip. The data for the curve is here and the script for processing is here.
Ariana will next work on the scripts to process surface area data. Symbiont cell density and total protein will be the next steps in processing fragments.
Surface area data was recorded in GitHub.
6 October 2022
Daily measurements
Ariana and Danielle took daily measurements of E5 tanks between 1000 and 1030. No mortality was observed. Daily measurements are recorded in GitHub here.
5 October 2022
Daily measurements
Ariana took daily measurements of E5 tanks between 1200 and 1230. No mortality was observed. Daily measurements are recorded in GitHub here. Ariana made more coated wire clips and will work on replacing these later this week.
Processing baseline fragments
Fragments that were airbrushed yesterday were removed from 10% bleach cups. Skeletons were clean and placed back in whirl pack bags. These will be processed for wax dipping to measure surface area. We will use the standard curve for this wax batch from the one generated by Danielle during this field trip. The data for the curve is here and the script for processing is here.
4 October 2022
Daily measurements
Ariana took daily measurements of E5 tanks between 1230 and 1300. Conditions were cloudy so light levels in all tanks were low (<200 PAR). The paper clips are continuing to rust, so these will need to be replaced soon with permanent coated wire clips. Ariana is working on making these permanent clips. No mortality is observed. Daily measurements are recorded in GitHub here.
Airbrushing baseline fragments
All fragments sampled during the baseline sampling on 2 October 2022 were airbrushed today between 1630 and 1830. Ariana airbrushed with the following proceedure:
- Remove 4 frags at a time from the freezer and store them in a cooler on frozen beads. Label 50 mL falcon tubes for each fragment. Falcon tubes were labeled with “E5 Apul Metabolism; Oct 2022 Baseline; Airbrushed; Putnam; Frag ID”.
- Each fragment was airbrushed with chilled PBS buffer into a zip bag. The tissue slurry in the bag was then poured into the respective falcon tube. The homogenate volume was recorded in GitHub here and written on the falcon tube.
- The homogenate was placed on ice until all 4 frags were completed.
- After airbrushing, the homogenate was put in the -40C freezer.
- The process was repeated for all 24 fragments.
- After airbrushing, all equipment was cleaned with DI water and alcohol wipes.
- Airbrushed skeletons were placed in plastic cups with 10% bleach overnight to clean the skeletons. These will be kept at room temperature until wax dipping to determine surface area.
Notes: Note that before aliquoting for assays, the homogenate will need to be homogenized after thawing. Additionally, some of the fragments were quite small, so homogenate volume was low (4-7 mL). We will need to decide which physiology metrics we would like to measure first to make sure we have enough homogenate.
Logger calibration
Ariana wrote a script to read the light and temperature logger calibration files from 25 September 2022. The script can be found here on GitHub. This script reads in files in a loop and then conducts a linear model for each logger to the standard. For temperature, we used the standard as the average of all black Hobo loggers. These loggers are higher resolution than the Hobo pendants. By using the average value of these loggers we can calibrate any offset between loggers as well as calibrate the pendant loggers to a standard. For light, the Hobo pendant loggers (measuring light in Lux) are calibrated to the Odyssey light logger (measuring light in Integrated PAR) to allow us to convert Lux to Integrated PAR for analysis.
This script outputs files with the logger serial number, coefficient, and intercept values. These files can then be read in when analyzing light and temperature data to conduct calibrations in later analyses.
3 October 2022
Tank cleaning
Ariana cleaned the E5 tanks today between 1100-1400. Tanks were cleaned by first removing the wire lines with frags attached from 1-2 tanks at a time and placing in the water table. The tanks were then removed and wiped with microfiber towels and rinsed with fresh water.
Fishing line of each fragment was cleaned and algae removed. Tanks were refilled and frags were added back in along with loggers and pumps. Ariana also cleaned the water lines.
During cleaning, tank position within each water table was rearranged. All tanks are in a new location. Frags were also returned such that each wire was put back in a different position to shuffle the fragments.
No mortality was observed in fragments. Water flow was adjusted to return to previous levels (350-400 mL per 5 sec).

Daily measurements
Daily measurements were taken between 1400 and 1500 and recorded in GitHub here. Conditions were cloudy and rainy today so light levels were low (<200 PAR).
2 October 2022
Daily measurements
Ariana took daily measurements at 1200-1230 in all E5 tanks. Recorded in GitHub here. Flow was even across tanks (350-375 mL per 5 sec) in each tank and did not need to be adjusted. Tomorrow the team will clean and rearrange all tanks according to the weekly maintainance protocol.
Baseline adult sampling
Ariana sampled adult fragments for baseline measurements as described in the baseline measurement preparation post on 30 September 2022. Sampling followed the baseline measurement protocol.
First, the selected fragments (N=24, n=4 per genotype) were removed from the tanks and imaged using a color standard and ruler for reference.

Fragments were then transported to the molecular lab for processing. As described in the protocol, each fragment was sampled for biopsies that were flash frozen (n=2 per frag) and stored in RNA DNA shield (n=2 per frag) with the remaining fragment frozen for physiology. Between samples, equipment was sterilized with 10% bleach followed by 70% ethanol. Gloves and counter were sterilized between each fragment. Sterilization was critical since these samples may be used for bacterial community characterization.
For each fragment, pre-labeled tubes and bags were used as described on 30 September 2022 post. Using bone cutters, 4 biopsies (scraping 5-10 polyp area) were taken and added to tubes. Flash frozen tubes were immediately frozen in liquid nitrogen. Tubes with RNA DNA shield were stored in the -40C freezer. The remaining fragment was stored at -40C. After all samplinig was completed, all tubes were then stored in the Gump molecular lab freezer at 40C.
Sampling of all fragments took 1.5-2 hours. Sampling of fragments was conducted in random order to avoid any time effects on genotype differences.
Checklists for sampling and sample inventory are available on GitHub.
Downloading loggers
HOBO Pendant loggers from the six E5 tanks were downloaded at 1220. Loggers were kept logging in the tanks. Black HOBO loggers from the wildtype parent tanks (n=2) were removed from tanks, stopped, and downloaded at 1030. Logger files can be found here.
1 October 2022
The team took the day off today for some fun snorkeling at Temae!
Daily measurements
Ariana took daily measurements of all tanks at 1630-1645. Sunlight was limited today and it was cloudy/rainy. Light measurements were all low during measurements (<25 PAR). Recorded in GitHub here. Flow was even across tanks (325-375 mL per 5 sec) in each tank and did not need to be adjusted. Ariana will check the outflows tomorrow and flush the lines to prevent lines clogging after sampling the adult baseline fragments.
30 September 2022
Daily measurements
Ariana took daily measurements at 0945-1020. Fragments look clean and healthy, no mortality observed. Flow adjusted to be between 350 and 400 mL per 5 sec in all tanks.
Daily measurements for today were recorded in GitHub here.
Preparing adult sampling
In the next couple of days Ariana will sample n=4 randomly selected fragments of each genotype to characterize baseline values of our responses of interest including DNA methylation and chromatin structure, gene expression, metabolomics, microbial communities, and physiology. Fragments were selected by randomly selecting n=4 per genotype such that each fragment was from a different tank and sampling from each tank was balanced to keep fragment density consistent.
Today, Ariana identified the fragments to be sampled and prepared and labeled all tubes, whirlpacks, and equipment that will be used.
Baseline sampling will be conducted following the protocol written by Ariana today. The baseline sampling protocol details procedures taking photographs of selected fragments for gene expression, microbial communities (16S and ITS2), chromatin structure and DNA methylation, metabolomics, and physiology.
The fragments that will be sampled are:
A7, 16, 20, 31
B9, 20, 25, 34
C1, 19, 26, 30
D4, 13, 26, 30
E5, 8, 20, 31
F2, 9, 17, 22
Labeling equipment
2 mL screw cap cryo tubes were labeled in preparation for sampling as nubmers 1-96. The number was written on the top cap of each tube as well as on the side. “E5 Metab. 2022” was also added on the side of each tube to identify the project. To see details on organization and labeling for baseline sampling, see the detailed protocol here.
Tubes #1-48 were filled with 1 mL of RNA DNA Shield. All tube ID’s and associated fragment and method of preservation were planned and recorded in the sample inventory.
Ariana also created a fragment tracking datasheet and baseline sampling checklist that will be used to track sampling and metadata for this project.
Two tube boxes were labeled as well with one box for RNA DNA shield samples (Tubes 1-48) and snap frozen samples (Tubes 49-96).
Finally, n=24 whirlpacks were labeled with fragment number as well as project information (“E5 Apul metabolism; Oct 2022 baseline; Putnam”).
Documents referenced in this post are all found below:
29 September 2022
Daily measurements
Ariana took daily measurements of the adult corals. The protocol for daily measurements can be found here.
The daily probe set up looks like the photo below. We take measurements of water flow, temperature (C), pH (mV), conductivity (psu), and light (PAR). Daily measurements except for light (see below) are taken at the center of each tank simultaneously.

Light is taken in the following locations in each tank:
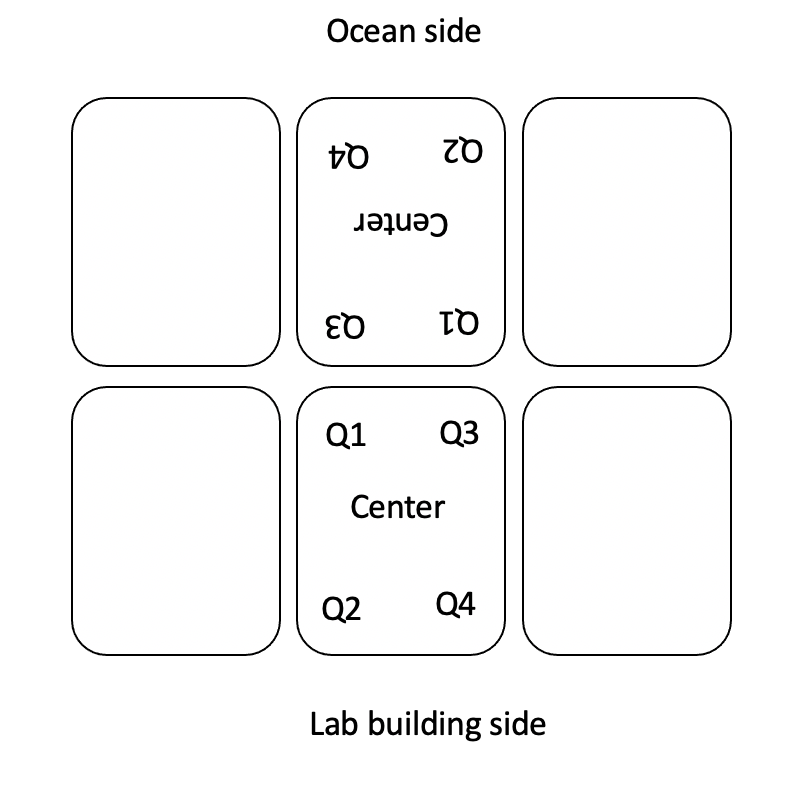
Daily measurements for today were recorded in GitHub here.
All tanks and fragments looked good today and no mortality was observed. Ariana is going to start making materials to replace the paperclips with coated wire. After about 1 week they will be rusting.
28 September 2022
Cleaning tanks
The team cleaned all large blue tanks today. Due to a clog in the line, water flow was cut off to one blue tank earlier this week resulting in some coral mortality. Dead portions of colonies were cut off of colonies and removed from tanks. All tanks were scrubbed and cleaned and filled with new water. Colonies will be taken back to the reef on Friday now that E5 fragmentation is complete (see below).
E5 adult coral metabolism experiment
Ariana cleaned all tanks, cleaned all fragments, secured clips to wire hangers, and finished labeling and fragmenting corals. Danielle assisted with fragmentation and labeling. These are described in more detail below.
Ariana has also started documents for entering data while tracking daily environmental measurements, conducting daily measurements and weekly cleanings, and tracking fragment survival in the GitHub repository. Ariana will write and post links to protocols here.
Fragmentation
Ariana finished fragmenting corals today for this project. There was high mortality of fragments for genotype C that was fragmented on 26 September 2022 (8 fragments showed mortality). Therefore, all C fragments were removed and replaced with a new genotype. This was also done for genotype A in which >80% of fragments showed signs of mortality. There was no other observed mortality in fragments from genotypes B, D, and E.
New genotypes for A and C fragments and for F fragments were fragmented at 1600-1800. 35 fragments were obtained from each genotype and were fragmented as previously described below. Fragments were attached to labels and added to tanks by 2100. The fragments varied in size from 2 cm to 6 cm in length depending on the available fragments from the morphology of the parent colony.
The final list of tank fragment locations is available on GitHub and listed below.
Tank 1:
- A1-A5
- B1-B5
- C1-C6
- D1-D6
- E1-E6
- F1-F6
Tank 2:
- A6-A11
- B6-B11
- C7-C11
- D7-D11
- E7-E12
- F7-F12
Tank 3:
- A12-A17
- B12-B17
- C12-C17
- D12-D17
- E13-E17
- F13-F18
Tank 4:
- A18-A23
- B18-B23
- C18-C23
- D18-D23
- E18-E23
- F19-F24
Tank 5:
- A24-A29
- B24-B29
- C24-C29
- D24-D29
- E24-E29
- F24-F29
Tank 6:
- A30-A35
- B30-B35
- C30-C35
- D30-D35
- E30-E35
- F30-F35
In the case that another genotype experiences mortality, there are 2 extra genotypes that were fragmented and will be held in the water tables.

Cleaning and tank set up
Ariana cleaned all treatment tanks by removing fragments and placing them in the water table with running water. Tanks were wiped clean and rinsed and re filled. Strings on fragments were cleaned to remove algae.
Ariana made notches in the hanging wires to prevent clips from moving around and the corals from getting too close to each other. These have worked well today.
In the next week, we will switch the paper clips to plastic clips to prevent rusting.


Feeding treatments
In 27 September 2022 post, Ariana described potential feeding treatments using artificial food. Ariana measured tank dimensions today for the E5 project - 40 cm W x 55 cm L x 30 cm H = ~66 liters. This is equivalent to about 18 gallons.
Reef Chili: Manufacturer recommends 1 scoop per 20 gallons. We would use 1 scoop per feeding per tank = 4 scoops per feeding total. If we assume 1 scoop = 1 gram, then we would have approx. 40 feedings per jar of reef chili (40 g per jar). If we feed 3x per week from mid-October to mid-December we will have 24 feedings. This will require One 1.4oz container has approximately 100 scoops. We should order 5 jars of reef chili to be safe. It can be stored in the fridge for up to a year if we do not use it. It looks like this item is currently out of stock.
Reef Roids: Manufacturer recommends 1 teaspoon per 100 gallons. We would therefore use 0.2 teaspoons per tank x 4 tanks = 0.8 teaspoons per feeding. With a total of 24 feedings, we would need ~20 teaspoons of Reef Roids. The bags are sold in units of 75 g or 150 g. On the website, it says “On average, the 30-gram bag of Reef-Roids will last roughly three months in a typical 50-gallon reef tank with bi-weekly feeding.”. If we calculate this out, bi weekly feeding of a 50 gallon tank for 3 months would use 0.5 teaspoons per feeding x 2 feedings x 12 weeks = 12 teaspoons in a 30 gram bag. If we need 20 teaspoons minimum, we would need to order the 75 g bag. To have extra, we should order the 150 g size. It can be refrigerated to last longer if we do not use it.
27 September 2022
Fragmentation
Ariana fragmented two more genotypes for use in the E5 adult experiment as described on 26 September 2022. Genotypes fragmented today were labeled D1-D35 and E1-E35. Fragments from colony A were showing mortality. These were removed from the tanks and will be replaced with a different genotype tomorrow. Fragmentation was completed from 0700-0900. Ariana and Danielle created labels in preparation for the final fragmentation tomorrow.
Fragments were then allocated to tanks with tank number recorded in GitHub here.
Fragments from yesterday appear to be healthy and the tank system is working well. In about a week we will need to change the paperclips to plastic coated paperclips to avoid rusting.
Once fragmentation is completed, Ariana will start daily measurements of environmental conditions, regular tank cleaning, and final treatment preparations.
The experimental design has been finalized for this project as is as follows:

This updated design includes a final processing for all fragments that includes the following responses:
- All fragments (5 per colony/genotype per tank) undergo CBASS testing at 5 temperatures including 27C, 29C, 31C, 32C, 33C (or something similar).
- We will take molecular samples from all fragments after CBASS.
- The fragment at the control temperature will then be used for molecular, metabolomic, and physiology responses.
Feeding treatments
For this experimental design, we need to plan for the type and amount of food to feed for Hollie to bring when she arrives in Moorea. Ariana looked in the literature for information on the effect of artificial foods (freeze dried plankton, rotifers, etc.) on coral growth.
Conlan et al. 2018: This paper compared coral growth with different types of food in A. millepora and P. acuta. They found no significant effect of feeding artificial foods on growth when compared to raw seawater. The dosage used was 0.05 g per tank in 49 L tanks. This equals about 1 mg per litre water.
Forsman et al. 2012: This paper examined the effects of different aritifical foods and dosage of foods on growth in M. capitata, P. acuta, and P. compressa. They tested 1X, 3X, and 10X of manufacturer’s dosage of Reef Roids, Reef Chili, and several other brands of food. Specifically, in M. capitata (in the Acroporidae family) has positive growth responses to artificial foods. They found that dosage of 1X of manufacturer’s instructions of Reef Roids or Reef Chili were best for this species. It is important not to overfeed, as this can reduce growth and cause tank algal overgrowth.
For feeding in this experiment, Ariana suggests that we purchase either Reef Roids or Reef Chili. We can feed corals 3X per week in treatment tanks. Water can be turned off for 1 h during feeding. Ariana will look into the amount of food that will be required for this feeding. If the amount is too high, we can look into building a feeding tank for all corals to be fed as a group and thereby reduce the amount of food needed.
Remaining to-do items for experiment set up
- Prepare a tank for 3x week feedings
- Remake paper clips to plastic clips
- Create a schedule document for daily measurements and weekly cleaning check offs
- Start treatment after ~ 2-week recovery period
- Order coral food
26 September 2022
Logger deployment
Ariana deployed pendant loggers for the E5 adult project. The following loggers were deployed. Tanks were numbered as 1-6 for simplicity.
| SSN | Interval | Time in Tanks | Tank |
|---|---|---|---|
| 21335989 | 10 min | 1115 | Tank 1 |
| 21002980 | 10 min | 1115 | Tank 2 |
| 21002972 | 10 min | 1115 | Tank 3 |
| 21335984 | 10 min | 1115 | Tank 4 |
| 20937873 | 10 min | 1115 | Tank 5 |
| 21002976 | 10 min | 1115 | Tank 6 |
| 20444033 | 10 min | 1115 | Wild parent tank 1 |
| 20719656 | 10 min | 1115 | Wild parent tank 2 |
Fragmentation
Today the team started the process of fragmenting adult A. pulchra corals for the E5 project to investigate the effects of altered metabolic state on epigenetic state and thermal performance.
To fragment, the team made and labeled strings (fishing line) attached to paper clips with lab tape as a label on the paper clip. These were then tied to coral fragments on the upper part of the branch such that the branch was oriented up right. The fragments were then clipped onto the wire using the paper clips. Fragments were suspended 15-18 cm below the water surface.
Coral fragments (~4-8 cm) were cut using bone clippers from larger colonies. Fragmentation was done in a water table from 1100-1900.

All treatment tanks were cleaned and pumps were added into each tank. Hobo pendant loggers are on the bottom center of each tank. Flow is high for these tanks to help corals recover from fragmentation. Note that no treatments have started yet, after fragmentation there will be a recovery period.


Fragments were then allocated to tanks with tank number recorded in GitHub here. Each tank has 5-6 fragments per colony and will have equal numbers of fragments.
A total of 3 genotypes/colonies were fragmented today and the team pre-labelled strings and paperclips for tomorrow. Ariana will finish fragmenting tomorrow and baseline sampling wll take place on Thurs/Friday of this week.
Fragmented today:
Colony A: Fragments A1-A35
Colony B: Fragments B1-B35
Colony C: Fragments C1-C25
Note that A fragments have some mortality. Ariana may switch out colony A for a different colony tomorrow. Water quality and flow was poor in this tank today that may have led to some of the mortality.
Tomorrow we will fragment colonies D, E, F.
Tank assignments
The tanks for each fragment are listed below and recorded in GitHub (see link above).
Tank 1:
- A1-5
- B1-5
- C1-6
Tank 2:
- A6-11
- B6-11
- C7-11
Tank 3:
- A12-17
- B12-17
- C12-17
Tank 4:
- A18-23
- B18-23
- C18-23
Tank 5:
- A24-29
- B24-29
- C24-29
Tank 6:
- A30-35
- B30-35
- C30-35
Revised E5 design
Given the amount of space in each tank and the number of fragments we could get per colony, Ariana re calculated our E5 design numbers. The changes are:
- Removing mid November time point. Since we want to know about how metabolic state influences stress response, it makes the most sense to keep our replication/samples for the time point in which we plan to do CBASS.
- Changed treatments to include autotrophy (light, no food), auto/hetero (light, added food), and heterotrophy (shaded, no food) as suggested by R. Cunning. This will provide nutritional treatments to shift metabolic state. Given our current design, we can do n=2 tanks per treatment.
- tanks will now include 5-6 fragments per colony. This will provide enough samples to take baseline samples this week as well as have 1 fragment for physiology and molecular and 3-4 fragments for CBASS. We can destructively sample following CBASS. The temperatures for CBASS will be chosen to represent a control, moderate heating, and high heating tempratures.
- Colony number has been reduced from 10 down to 6. There is not enough room in the tanks for all 10 colonies.
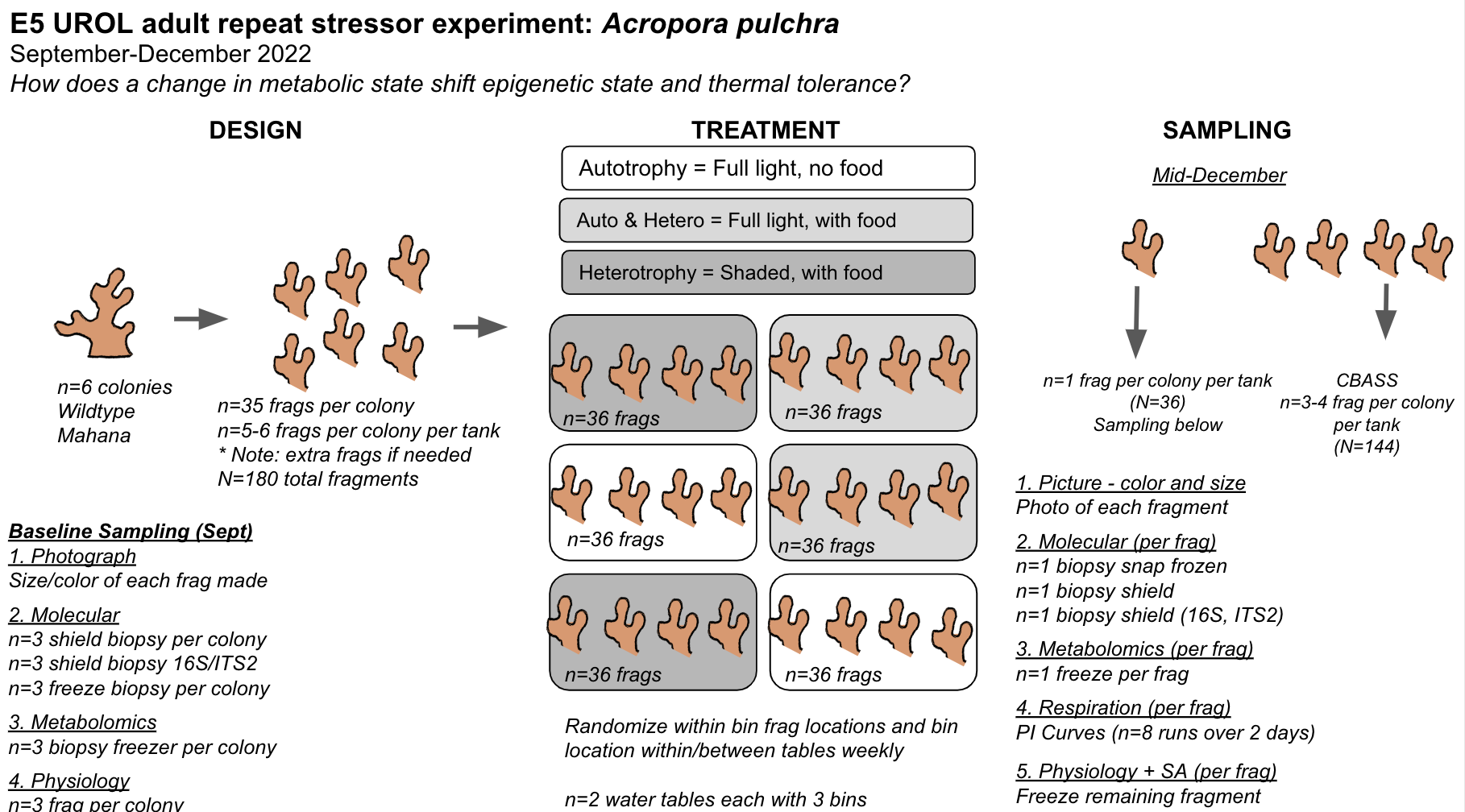
25 September 2022
Logger calibration
Ariana removed loggers from tanks at 1030 and downloaded all logger files at 1200 and began writing a script to calulate the offsets for each logger.
Tank set up
Ariana set up the E5 tanks to have hanging wires for each fragment when fragmentation occurs tomorrow.


Planning for tomorrow
Tomorrow Ariana will fragment the A. pulchra corals. These are the steps we will follow to fragment:
- Select corals with the most fragments/branches (n=10 preferred).
- Place the first colony in a holding tank on the water table.
- Use bone cutters and a frag saw to cut as many fragments as possible approx. 3-4 in long at the branches.
- Label the fragments by attaching to a paperclip with a tape label using fishing line. Label with a colony letter and number (e.g., A1-A30, B1-B30, etc.).
- Place the fragments in the tanks randomly distrubted between the tanks.
- Return the remaining colony to the holding tank.
This fragmentation will be followed by baseline sampling at the start of the treatment period depending on the number of fragments available.
24 September 2022
Logger calibration
Ariana added the two pendant loggers from the incubators into the calibration tank that were missed yesterday.
Logger 21335989 - logging every 10 min
Logger 21002976 - logging every 10 min
Added to calibration tank at 0900. Note that data from 0700-0830 may need to be removed from the calibration. At this time the morning sun is causing shadows in the tank that may make the calibration in accurate.
Daily measurement probe set up
Daily measurements for larval and the E5 adult project should include:
- Flow rate (mL / 10 sec): Use a graduated clyinder to measure the amount of water from the input tube in a 10 sec period.
- Temperature (C): Measurement of temperature in the center of each tank.
- Light (PAR): Measurement of light at the center of the tank and in the center of each of 4 quadrants (total of 5 measurements per tank).
- pH and salinity: Measure pH and salinity with the Orion probe at the center of each tank.
Time of day, tank number, and notes regarding weather or other conditions should be noted.
For Ariana’s larval project, daily measurements will be recorded in Google Drive here.
For the E5 adult project, daily pH, temperature, and flow measurements will be recorded in GitHub here and daily light measurements will be recorded in GitHub here.
The protocol for taking daily measurements was written by Ariana available here in the E5 protocols repository based on the Putnam Lab protocol.
Ariana prepared all probes and measured a standard curve for the pH probe across a range of temperature with Tris using the Putnam Lab protocol. 15 mL of Tris was added to a falcon tube and placed in the fridge for ~20 min. The temperature and pH probe were then submerged into the falcon tube on the bench top. The calibration started at 11°C and slowly warmed to room temperature with measurements of temperature and Tris every ~1°C. Once the Tris reached room temperature the Tris and probes were moved into a benchtop incubator (MyTemp Mini) and warmed to the upper temperature used in the calibration (~35C).
This data is below and was recorded in Ariana’s Google Drive here and in the E5 repository here.
| Date | Time | Temp.Tris | mV.Tris |
|---|---|---|---|
| 20220924 | 1013 | 11.18 | -73 |
| 20220924 | 1016 | 13.31 | -72 |
| 20220924 | 1020 | 14.66 | -70.3 |
| 20220924 | 1025 | 16.18 | -68.3 |
| 20220924 | 1030 | 17.28 | -66.7 |
| 20220924 | 1049 | 18.75 | -64.9 |
| 20220924 | 1057 | 19.48 | -64.1 |
| 20220924 | 1102 | 20.18 | -63.1 |
| 20220924 | 1106 | 21.08 | -61.9 |
| 20220924 | 1111 | 22.19 | -60.6 |
| 20220924 | 1115 | 23.07 | -59.4 |
| 20220924 | 1120 | 24.04 | -58 |
| 20220924 | 1125 | 25.03 | -56.7 |
| 20220924 | 1130 | 26.06 | -55.4 |
| 20220924 | 1135 | 27.27 | -53.9 |
| 20220924 | 1140 | 28 | -52.8 |
| 20220924 | 1145 | 28.96 | -51.8 |
| 20220924 | 1150 | 30.42 | -49.6 |
| 20220924 | 1153 | 31.06 | -48.6 |
| 20220924 | 1159 | 32.27 | -47.1 |
| 20220924 | 1204 | 33.35 | -45.6 |
| 20220924 | 1210 | 34 | -44.8 |
| 20220924 | 1215 | 35.12 | -43.1 |
| 20220924 | 1220 | 36.22 | -41.6 |
23 September 2022
Project set up
Ariana cleaned water lines and started water in n=6 tanks in preparation for the adult experiment.
Ariana started a GitHub repo for this project here.
Logger offload and calibration
Loggers were read off for the wildtype parent tanks and Danielle’s parent tanks. Files were labeled with SSN and date and saved in the Apulchra_metabolism E5 GitHub repository. Loggers read off were:
20719656 (Wildtype Tank 2)
20946644 (Danielle Tank 1)
20444033 (Wildtype Tank 1)
20444040 (Danielle Tank 2)
The wildtype colonies are those that will be fragmented and used for this experiment.
Ariana started a calibration of all temperature and light loggers that will run over the next 24-48 h. This will allow us to convert Lux to PAR from the Hobo Pendant loggers and to calculate temperature offsets from the temperature logging on the Hobo Pendants.
The following loggers were launched:
| Logger | Interval | Type | Time Deployed |
|---|---|---|---|
| 20719656 | 10 min | Hobo Temp | 1635 |
| 20946644 | 10 min | Hobo Temp | 1635 |
| 20444033 | 10 min | Hobo Temp | 1635 |
| 20444040 | 10 min | Hobo Temp | 1635 |
| 21335986 | 10 min | Pendant | 1635 |
| 21002981 | 10 min | Pendant | 1635 |
| 21002972 | 10 min | Pendant | 1635 |
| 20937872 | 10 min | Pendant | 1635 |
| 21002975 | 10 min | Pendant | 1635 |
| 21335980 | 10 min | Pendant | 1635 |
| 21335987 | 10 min | Pendant | 1635 |
| 21002980 | 10 min | Pendant | 1635 |
| 21002977 | 10 min | Pendant | 1635 |
| 21002974 | 10 min | Pendant | 1635 |
| 21335982 | 10 min | Pendant | 1635 |
| 21002982 | 10 min | Pendant | 1635 |
| 20937873 | 10 min | Pendant | 1635 |
| 21335983 | 10 min | Pendant | 1635 |
| 21335984 | 10 min | Pendant | 1635 |
| 21002979 | 10 min | Pendant | 1635 |
| 15643 | 10 min | Odyssey Light | 1635 |
The Odyssey logger was previously calibrated by Danielle Becker-Polinski with this protocol.
Loggers were arranged as pictured below in one tank with running water at a low flow to avoid surface disturbance. These loggers will run for 24-48 h to conduct the calibration for light and temperature data.

22 September 2022
Project set up
Ariana constructed tanks from the 6 plastic bins that were purchased yesterday for the experiment. Holes were drilled in the top edges of the bins for wires to be secured to hang coral fragments and a drain hole was added.
Tomorrow, Ariana will add water flow to these tanks and prepare materials for sampling and fragmentation over the weekend.
21 September 2022
Project planning
Ariana discussed experimental design with the E5 team and we have revised the experimental approach as pictured below.
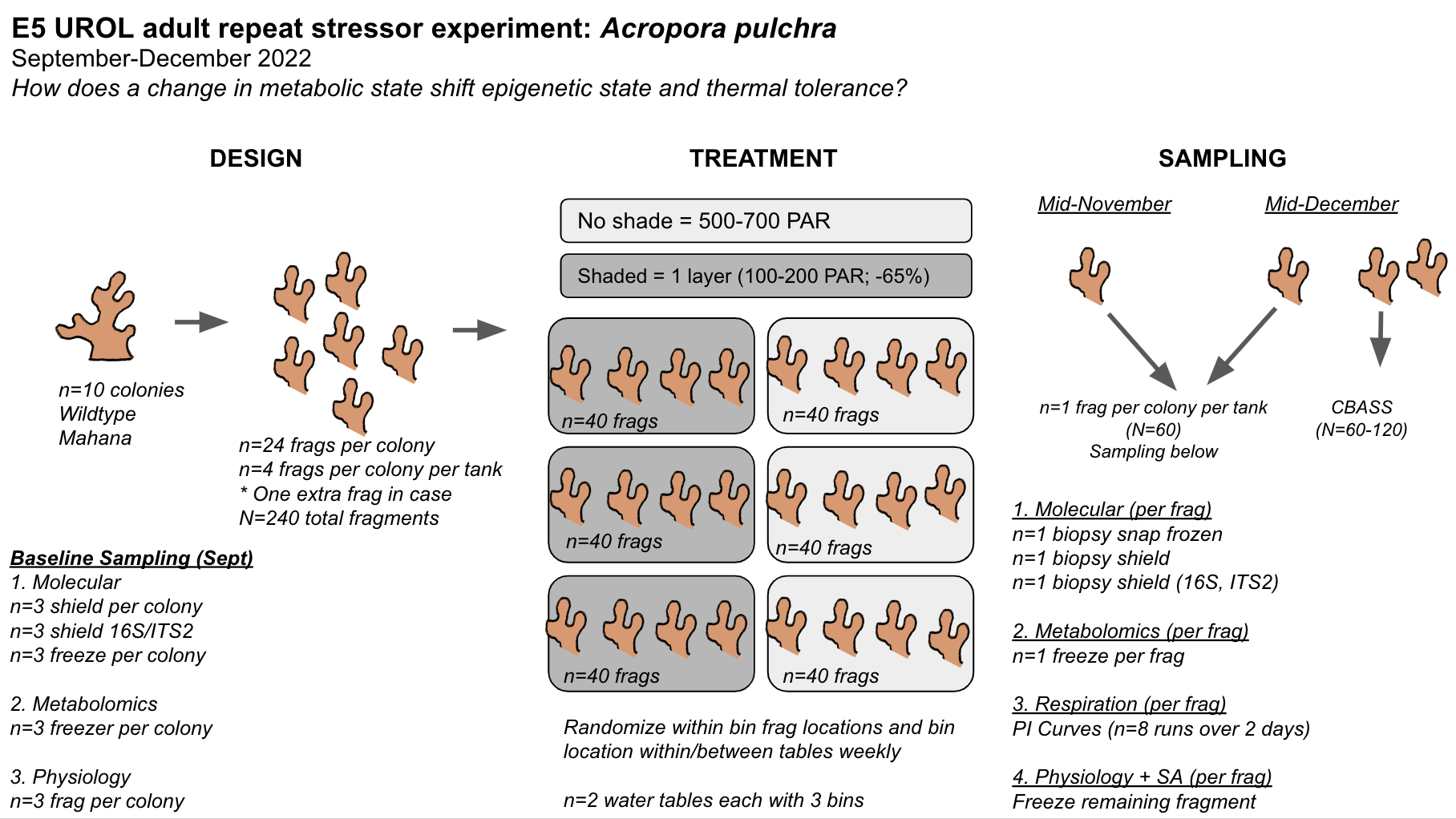
This revision includes these updates:
- Moving sampling time points from 3 (Oct, Nov, Dec) to two time points (Nov and Dec) to maximize the number of fragments per colony needed with available material.
- Added biopsy for ITS2 and 16S sequencing during sampling.
- Increase number of genotypes/colonies to n=10
- We will now need n=3 fragments per colony per tank in order to sample one fragment per colony at each time point. We will try to obtain n=3-5 fragments per colony per tank in the case that we want to increase sampling for CBASS and having an extra fragment in case of mortality.
We will continue to talk with the E5 team to finalize the design prior to fragmenting and starting the experiment early next week.
Project set up
Ariana, Pierrick, and Danielle sourced materials for the E5 adult project including buying tank bins, finding shade cloth, and cleaning water tables. We have black shade cloth with rebar on either side that fit on our tank systems. We purchased 6 plastic bins that will be used as tanks. We cleaned water tables to prepare space for these tanks.
Ariana measured light levels under ambient conditions (under covered tank area with no shade cloth) and under two low light conditions (1 layer of shade cloth and 2 layers of shade cloth). Light levels in the high light treatment will be 500-700 PAR peaking during the day. Under one layer of shade cloth, light was reduced by 65% to approx. 100-300 PAR. Under two layers of shade cloth, light was reduced 80% to <100 PAR. We will use one layer of shade cloth for this experiment to provide sufficient light for coral growth in both treatments.
We found recirculating pumps for each tank. We also prepared equipment for hanging the fragments. We will drill holes across the length of each tank to secure wire. We will then use paper clips to secure fragments tied to fishing line to hang fragments in the tank. This will help reduce algal growth and will be more efficient to set up and maintain.
Ariana will finish set up for this project this week and then move forward with fragging and starting the experiment once design is finalized.
Boating
Danielle gave the team an orientation to driving the LTER boat (509) at Gump. Ariana took 4 trips as captain driving in the channels to learn navigation on the north shore. Ariana and Pierrick will work on obtaining their authorization for driving Gump boats.
20 September 2022
Project planning
Ariana developed a draft experimental design for the adult coral E5 repeat stressor experiment. Ariana conducted planning and sourcing materials for this project today.
Overall, this project will expose coral colonies to ambient (450-600 PAR) and reduced (100-200 PAR) light conditions. A draft design is included below. This experiment will run until December. Monthly, fragments will be sampled for molecular, metabolomic, and physiological metrics. In December, S. Matsuda and R. Cunning will measure thermal phenotypes using the CBASS system.
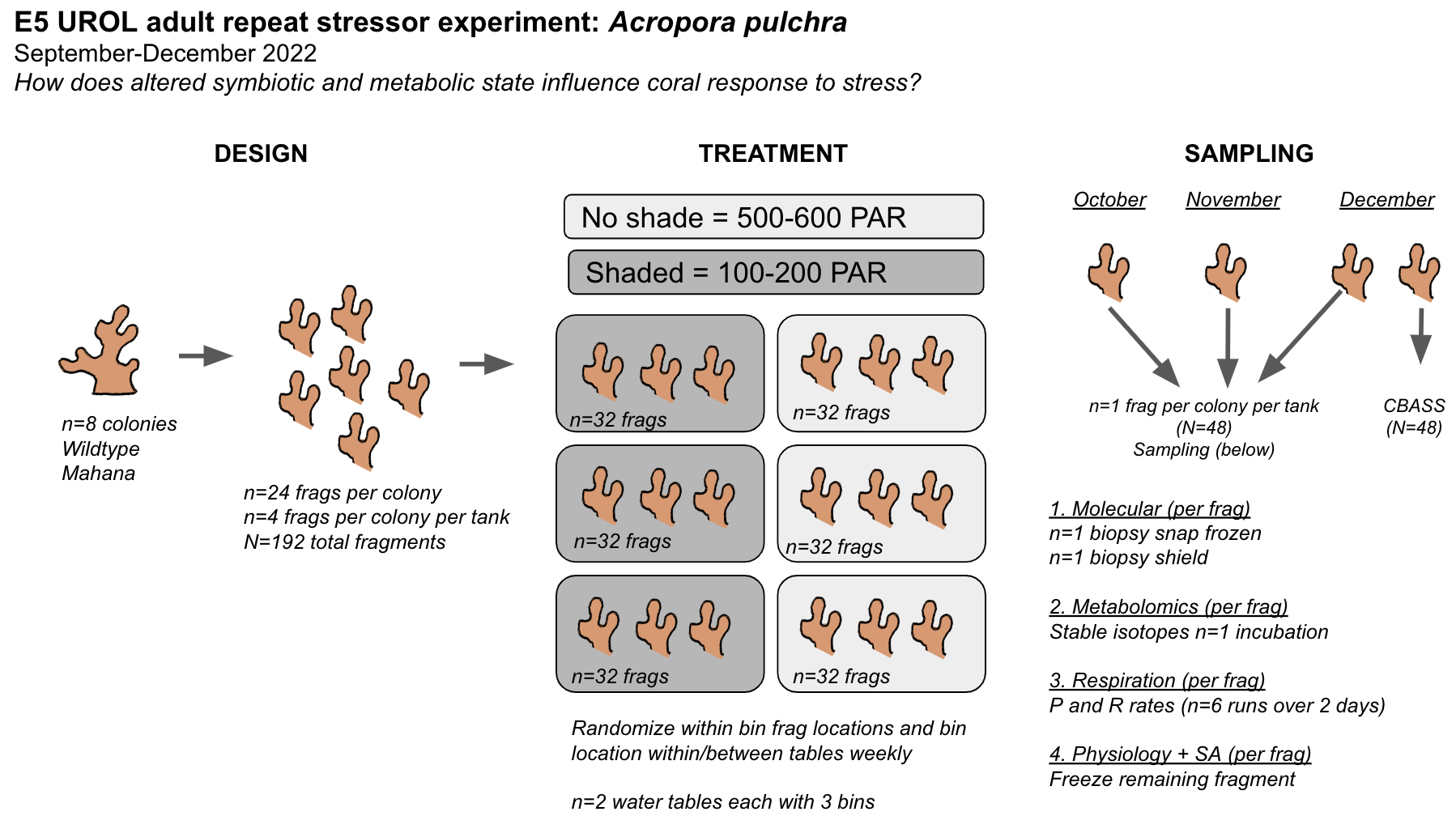
The parent colony collection and maintenance is described in our daily entry notebook. Parent colonies were collected from the Mahana site on Moorea’s north shore (17°29’12.5”S 149°53’16.1”W) on 16 September 2022.

We collected n=25 colonies (approx. 15-30cm in diameter) from large patches of Acropora pulchra from the Mahana field site between 1000-1200. We have a year-long project that has been tracking histology and physiology of colonies (Danielle’s project) at this site and therefore we have temperature and light data from this site as environmental context. The colonies collected are considered “wildtype” and were not taken from tagged colonies. Colonies were collected from different patches to obtain a genetically diverse sample.
Ariana and Danielle used a hammer and chisel to remove large fragments (15-30cm diameter) from these patches. Ariana Danielle and Pierrick transported them back to the boat where they were kept in coolers and buckets filled with seawater. Colonies were then transported back to Gump station at 1230 and placed in 2 flow through seawater tanks (n=12-13 colonies per tank). Tanks are equipped with 3 small recirculating pumps for water flow and had high water flow.

These colonies were monitored for spawning in September but did not spawn.
